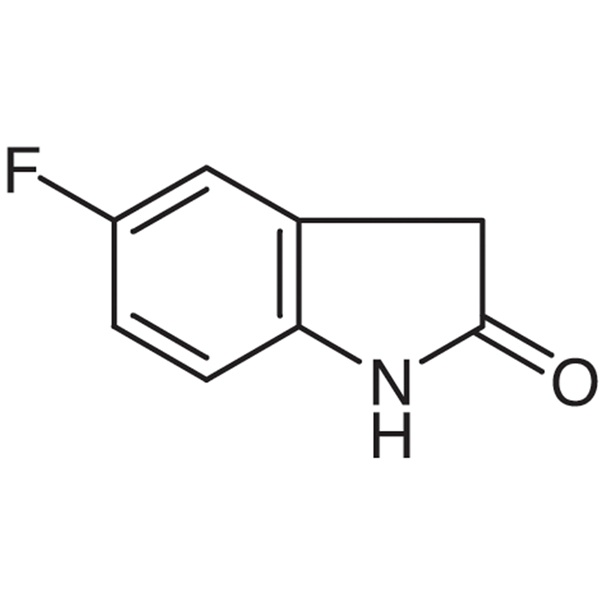
5-Fluoro-2-Oxindole CAS 56341-41-4 Purity >99.0% (LCMS) Sunitinib Malate Intermediate Factory
Shanghai Ruifu Chemical Co., Ltd. is the leading manufacturer and supplier of 5-Fluoro-2-Oxindole (CAS: 56341-41-4) with high quality, commercial production. Welcomed to order.
| Chemical Name | 5-Fluoro-2-Oxindole |
| Synonyms | 5-Fluorooxindole; 5-Fluoro-2-indolinone; 5-Fluoroindolin-2-one; 5-Fluoro-1,3-Dihydro-indol-2-one |
| CAS Number | 56341-41-4 |
| CAT Number | RF-PI1543 |
| Stock Status | In Stock, Production Scale Up to Tons |
| Molecular Formula | C8H6FNO |
| Molecular Weight | 151.14 |
| Density | 1.311±0.06 g/cm3 |
| Brand | Ruifu Chemical |
| Item | Specifications |
| Appearance | Light Yellow Crystalline Powder |
| 1 H NMR Spectrum | Consistent With Structure |
| LCMS | Consistent With Structure |
| Purity / Analysis Method | >99.0% (LCMS) |
| Melting Point | 143.0~147.0℃ |
| Loss on Drying | <0.50% |
| Total Impurities | <1.00% |
| Test Standard | Enterprise Standard |
| Usage | Pharmaceutical Intermediates; Sunitinib Malate Intermediate |
Package: Bottle, Aluminium foil bag, 25kg/Cardboard Drum, or according to customer's requirement.
Storage Condition: Store in sealed containers at cool and dry place; Protect from light and moisture.


5-Fluoro-2-Oxindole (CAS: 56341-41-4) is mainly used in pharmaceutical industry as a pharmaceutical intermediate. 5-Fluoro-2-Oxindole is an intermediate in the synthesis of Sunitinib Malate (CAS: 341031-54-7). Sunitinib Malate is a kind of novel oral multi-targeted anticancer drugs and belongs to multi-targeted tyrosine kinase inhibitor with its trade name being “Suntent”. It was successfully developed by Pfizer Company and has dual anti-tumor effect. Moreover, it is the only therapeutic drug which can go beyond the 2-year survival period of advanced kidney cancer and plays a central role in the field of carcinoma and gastrointestinal stromal tumor therapeutic areas. It entered into market in February 2006 in the United States. This drug was the first anti-cancer drug which was approved by the US FDA and can simultaneously treat two diseases.
-
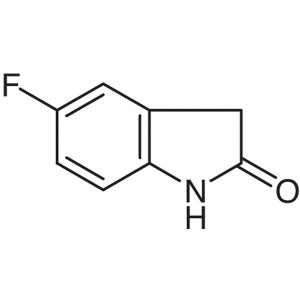
5-Fluoro-2-Oxindole CAS 56341-41-4 Purity >99.0...
-
5-Hydroxyoxindole CAS 3416-18-0 Purity ≥95.0% H...
-
6-Chlorooxindole CAS 56341-37-8 Purity >99.0% Z...
-
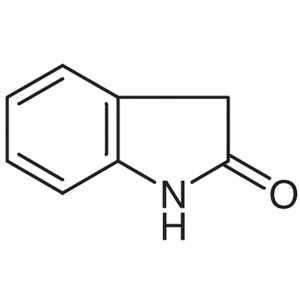
Oxindole CAS 59-48-3 Purity >99.0% (HPLC) Facto...
-
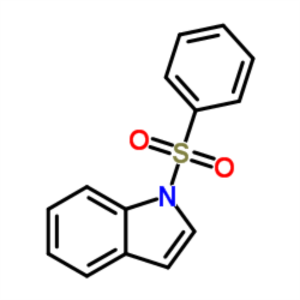
1-(Phenylsulfonyl)indole CAS 40899-71-6 Purity ...
-
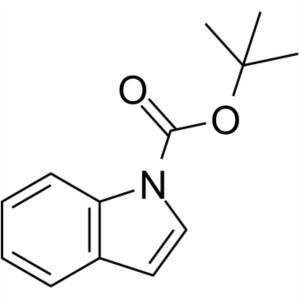
1-Boc-Indole CAS 75400-67-8 Purity >96.0% (HPLC)
-
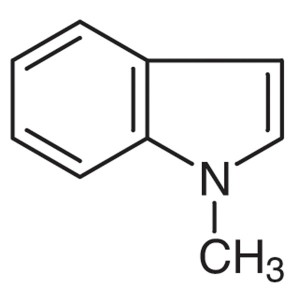
1-Methylindole CAS 603-76-9 Purity >98.0% (GC) ...
-
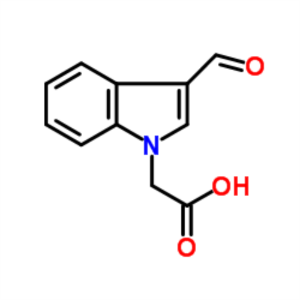
1-Methylindole-3-Carboxaldehyde CAS 138423-98-0...
-
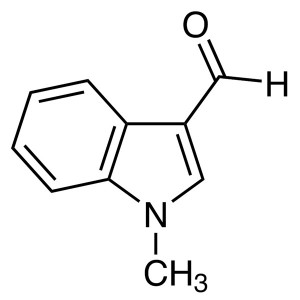
1-Methylindole-3-Carboxaldehyde CAS 19012-03-4 ...
-
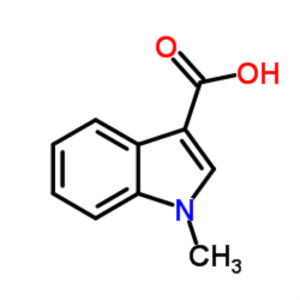
1-Methylindole-3-Carboxylic Acid CAS 32387-21-6...
-
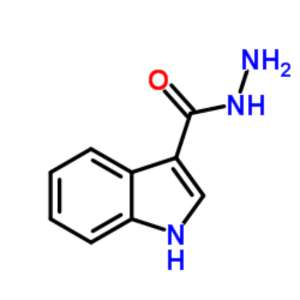
1H-Indole-3-Carbohydrazide CAS 15317-58-5 Purit...
-
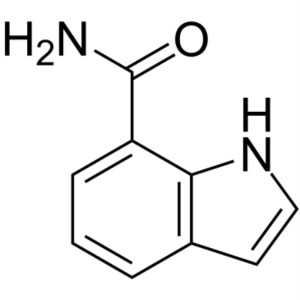
1H-Indole-7-Carboxamide CAS 1670-89-9 Purity >9...
-
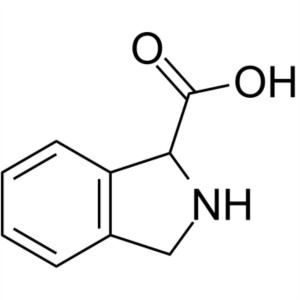
2,3-Dihydro-1H-Isoindole-1-Carboxylic Acid CAS ...
-
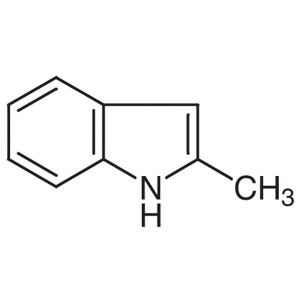
2-Methylindole CAS 95-20-5 Purity >99.0% (GC) F...
-
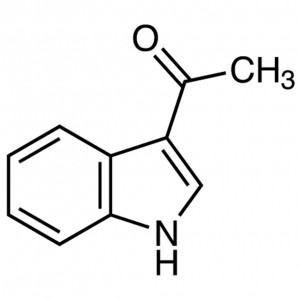
3-Acetylindole CAS 703-80-0 Purity (LCMS) ≥98.0...
-
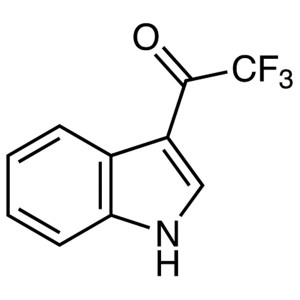
3-(Trifluoroacetyl)Indole CAS 14618-45-2 Purity...